H-ABC / TUBB4A Leukodystrophy Community BLOG
Building a Rare Disease Community: Why Connection Supports Progress
Connection within the rare disease community plays an important role in supporting both care and research, particularly for conditions such as H-ABC/TUBB4A related leukodystrophy where individuals may be geographically dispersed and limited in number. Advocacy organizations, including the Foundation to Fight H-ABC/TUBB4A, help facilitate these connections by sharing information, supporting participation in registries and studies, and fostering collaboration among families, clinicians, and researchers. Through these coordinated efforts, community engagement contributes to a broader understanding of the condition and helps support a more informed and connected approach to ongoing research and care.
Speech and Communication in H-ABC/TUBB4A Related Leukodystrophy: What We Know and What We’re Learning
Communication development in H-ABC/TUBB4A related leukodystrophy is influenced by changes in white matter that can affect motor control and the coordination required for speech. As a result, individuals may experience a range of communication differences, from early delays to changes over time, reflecting both the variability of the condition and its neurological basis. Approaches such as speech-language therapy and augmentative and alternative communication (AAC) can support functional communication, while ongoing research is increasingly recognizing communication as an important measure of daily function and quality of life.
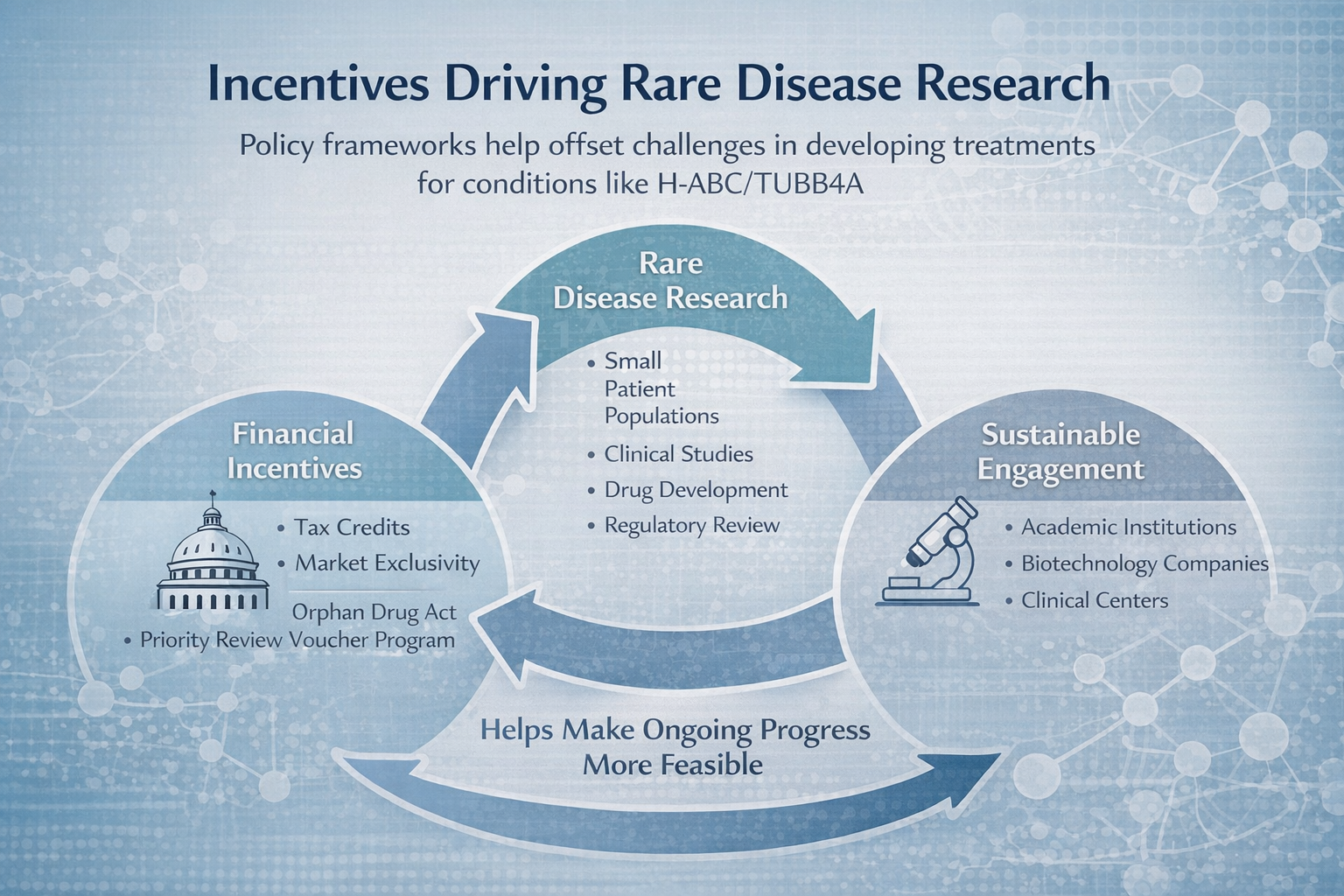
The Role of Incentives: How Policy Shapes Rare Disease Innovation
Policies such as the Orphan Drug Act and the Rare Pediatric Disease Priority Review Voucher program play an important role in supporting rare disease research by helping to offset the financial and logistical challenges associated with developing therapies for small patient populations. For conditions such as H-ABC/TUBB4A related leukodystrophy, where the number of identified individuals is limited, these incentives can influence whether research programs move forward and how resources are allocated. While they do not remove the complexities of drug development, they contribute to a framework that makes continued scientific progress more feasible and helps sustain engagement across research, clinical, and industry communities.
Natural History Studies: Why They Matter Before a Cure Exists
Natural history studies play an important role in rare disease research by helping to build a clear understanding of how a condition develops over time. In diseases such as H-ABC/TUBB4A related leukodystrophy, where relatively few individuals have been identified worldwide and clinical presentation can vary, this type of longitudinal observation provides essential context for interpreting change. By documenting how symptoms progress in the absence of treatment, natural history data helps inform clinical trial design, supports the identification of meaningful outcome measures, and contributes to a more structured path toward future therapeutic development.
The Diagnostic Journey: Why Rare Diseases Still Take Years to Identify
The path to a diagnosis for H-ABC/TUBB4A and other rare diseases is often long and complex, with families frequently waiting years, seeing multiple specialists, and facing misdiagnoses before reaching answers. With an average delay of 4–6 years and most rare diseases being genetic, timely access to testing remains a critical challenge, impacting care and support. Earlier diagnosis can significantly improve outcomes by connecting families to appropriate care, resources, and research opportunities, making continued efforts to strengthen diagnostic pathways essential.
Federal Incentive for Rare Pediatric Disease Therapies Extended Through 2029
Hope for Rare Disease Families: Congress Extends Pediatric Drug Incentive Program
Congress has extended an important federal incentive that supports the development of treatments for children with rare diseases. Through the Consolidated Appropriations Act, 2026, lawmakers renewed the Rare Pediatric Disease Priority Review Voucher program through 2029. The program allows the U.S. Food and Drug Administration to award valuable priority review vouchers to companies that successfully develop therapies for rare pediatric diseases.
The Ripple Effect of Rare
On the last day of February, Rare Disease Day commands global attention — shining a light on conditions most people will never encounter, yet millions of families confront every single day.
This is not merely a date on the calendar. It is a declaration that rare does not mean invisible. It is a reminder that awareness, while vital, is only the beginning. Real progress demands relentless research, courageous advocacy, and an unwavering commitment to ensure that no life is diminished or delayed simply because a diagnosis is uncommon.